Instrument Snapshot
The Bruker BioSpec 3T MRI features an 8.2 cm inner diameter RF coil for large rat studies, complete with a motorized animal positioning system for routine handling and increased throughput with automated multistage, whole-body imaging.
With over 100 validated and ready-to-use in vivo protocols and scan programs for mice and rats, this high-resolution imaging instrument will provide clear in-vivo images for your studies.
Reach out to our core facility in Boston with any questions at cils-core@northeastern.edu.
Features
Automated animal positioning
Mulitmodal rat/mouse cradle
Localized MR Spectroscopy
Sample Images Acquired with Body Volume Coils
Application: Control Rat- Abdomen
Sequence: Bruker:RARE
Repetition/Echo Time: 925/55 ms
Resolution: 1 mm x (313×313) µm2
Comments: Respiratory Gating, 5 min
Courtesy of Bruker
Application: Control Mouse – Cardiac
Sequence: Bruker:IgFLASH
Repetition/Echo Time: 20/3.12 ms
Resolution: 1 mm x (125×125) µm2
Comments: Flip Angle = 20°, 10 min
Courtesy of Bruker
Application: Control Mouse – Lungs
Sequence: Bruker:UTE3D
Repetition/Echo Time: 2.7/0.008 ms
Resolution: (192x192x385) µm2
Comments: Flip Angle = 3.5°, 10 min
Courtesy of Bruker
Rates
Internal NU
$30/hr
External Academia
$150/hr
Industry
$200/hr
Bruker BioSpec 3T Brochure
PET/MR 3T
Summary of MRI Key Performance Parameters
FAQs
General Questions
What is MRI?
Magnetic Resonance Imaging (MRI) is a non-invasive imaging technique that utilizes a combination of a strong magnetic field and radiofrequencies to examine soft-tissues (brain, breast, spinal cord, ligaments, cartilage…) with high-penetration depth.
How does MRI work?
Animal bodies are mainly composed of hydrogen atoms (“protons”). These protons spin randomly under normal circumstances. When placed under a strong magnetic field, the protons align in the same direction. Radiofrequency waves can be transmitted through the tissues resulting in signal from the protons, which can be measured and processed to calculate an MR image.
What is the resolution of the scanner?
The short answer is “it depends”.
MRI is an imaging technique “truly three-dimensional (3D)” and resolution in MRI is dependent on the interplay of three main factors: signal per voxel, spatial resolution, and time.
- Signal per voxel depends on the size of the voxel and the application of gradients, but it is limited by the number of protons within that voxel. Signal to noise ratio (SNR) within a voxel increases with the size of the voxel, number of signal averages (NSA), the number of encoding steps (Nphase, Nfreq), and decreases with increasing receiver bandwidth (RBW).
- Spatial resolution (SR) is inversely proportional to the voxel volume, which can be calculated by multiplying the slice thickness (ST) by the field-of-view (FOV) and dividing by the matrix size (MS) such that SR = ST * FOV / MS.
- Time/temporal resolution defines the physical time that is necessary to acquire a single scan.
To increase the spatial resolution, we could reduce the voxel volume, say, 8-fold. However, this would lead to a decrease in signal per voxel (and SNR). In efforts to compensate for the decrease in voxel volume, we would need to increase the NSA by 8-fold, which would result in an increase in scan time of 28 = 64-fold, completely undesired.
For additional information, take a look at: Summary of Key Performance Parameters
What are the main MRI applications?
embed here
Animal-Related Questions
I want to image animals, what should I do?
- Project approval by the director of the facility
- Safety training completed (BIOraft)
- Animal protocol approved (IACUC guidelines)
- MRI training by staff members of the facility successfully completed
- Reservation in FBS completed
Do I need to follow IACUC guidelines?
Absolutely!! Users need to develop and amend their own IACUC protocol to include any imaging procedures in addition to room information. Descriptions of imaging methodologies and protocol templates are available upon request.
What should I add in the IACUC protocol?
For imaging studies without invasive procedures, we recommend adding a paragraph similar to the following example for mice in the Experimental Design section:
“The scope of this study will be limited to imaging of _____strain & animal____ without any invasive procedures. MRI is a non-invasive technique and generally regarded safe to animals. Imaging will be performed on the following parts of the animal: ______heart/brain/limbs/internal organs…____. No invasive procedure will be performed before or during the imaging
Mice will be anesthetized with 1-3% isoflurane and placed in an animal cradle with integrated temperature control and ECG/respiration sensors in the MRI scanner. Each MRI scan will take around ____ minutes under different MRI sequences. After imaging is complete, the mouse will be monitored for recovery (ambulation, maintenance of sternal recumbency) from anesthesia before being returned to the animal facility. “
If surgery procedures or more advanced imaging protocols are needed, please contact us (g.rong@northeastern.edu) or DLAM (dlam@northeastern.edu)
What animals or samples can I image in the scanner?
We currently have two volume coils, one for rat and one for mouse, 60- and 30-mm inner diameters. Typically sized animals perfectly fit. Most samples and animals smaller than these measurements can be imaged in the scanner. There are also 3D printed holders for in-vitro imaging specifically designed to hold up to 13 PCR tubes and a microscope slide.
Is there a limit in sample/animal size? How big can my sample/animal be?
The subject must fit within the coil, limited to 30 mm diameter (mouse coil) and up to 60 mm diameter (rat coil). Slice dimensions can then be defined for each MRI acquisition independently. In general, it is important to keep the subject as centered as possible within the magnet, referred as “isocenter”, which has the most homogeneous magnetic field distribution. If the area you want to image is bigger than the largest possible FOV, you can use Image Stitching – see specific Protocol for more details.
Is there any lab space for animal preparation before or after the imaging study?
Yes, there is an adjacent room with available space for animal preparation as well as tissue collection procedures. Please discuss with the core director if you are planning on using these spaces for long periods of time (> 30 minutes) before or after the imaging experiments. These rooms MUST be included in the approved animal protocol before using them.
Does the animal need to be restricted during imaging?
Ideally, the animal should be as still as possible, to prevent any undesired artifacts in the image (movement causes blurring).
How can I avoid artifacts from the animal body motion?
We suggest restricting the animal as much as possible to ensure that it does not move during the scan, without compromising its well-being. In addition, we recommend using respiratory and ECG-gating (either retrospective or prospective) so that images are acquired in synchrony with the physiological motion, in efforts to minimize these motion-related artifacts.
Do I need to bring my own anesthesia supply?
We offer inhaled isoflurane administered in a mixture of oxygen, in both the MRI prep room and the imaging area.
Do I need to bring my own contrast agent for imaging?
Yes, users need to bring their own contrast agent, which MUST be included in the animal protocol.
Can I bring animals back to the animal housing facility?
Generally yes, but please discuss with DLAM before initiating your animal experiments for confirmation.
Safety-Related Questions
Is MRI safe?
MRI is non-ionizing and under proper usage, it is safe/harmless for operators. However, the powerful magnet can cause attraction of metallic objects in close proximity.
MRI can be unsafe to you if there are non-MRI safe objects that can result in a dangerous situation. Examples include: metallic objects (scissors), pacemakers, metal particles in eyes, medication patches, heart and/or brain surgery, foreign objects inside your body (surgical clips or staples), some tattoos, implants (including Cochlear, Nerve Stimulator (Tens Unit), drug pump, Cardioverter-Defibrillator (ICD)…)
Please discuss with the Core director any of these concerns to ensure safety.
Will I be exposed to radiation or any side effects?
No- this scanner does not emit radiation and has no known side effects. However, it is important to be careful with any metal objects due to the strong magnetic field.
What can I (not) wear/have with me while at the MRI facility?
Lab coat, safety glasses, gloves (general PPE) is required.
Food and drinks are not permitted even in the control room.
Please avoid carrying the following non-MRI compatible items, even in your pockets: Hearing Aids, Wallets, Credit/Debit Cards, Jewelry, including Body-Piercing Jewelry, Keys, Watches, Loose Change, Eyeglasses, Cell Phones, Pagers, PDA´s, Storage Media, Tablets/Laptops/Computers, Dentures, Prosthetic Devices, Insulin Pumps, Hair Pins/Bobby Pins.
Why can I not carry non-MRI compatible items?
The MRI scanner has a powerful magnet that can damage (even completely destroy) hearing aids, watches, cell phones, PDA´s, storage media, insulin pumps, electronic keys, etc. Additionally, MRI magnet can erase credit/debit cards, launch metallic objects as projectiles (which become hazardous). Lastly, the effects of any of these objects in proximity to the magnetic field can seriously damage/degrade image quality.
Questions About Contrast Agents (CA)
What is an MRI contrast agent?
MRI contrast agents are substances designed to facilitate the visualization of certain tissues of interest. Gadolinium-based contrast agents are the most common in MRI applications (e.g., Gadovist, Dotarem), and are FDA approved for clinical settings.
When is a contrast agent needed?
MRI contrast agents are utilized to facilitate visualization of certain tissues. It can be administered intravenously, intraperitoneally, subcutaneously, etc. Any administration of contrast agent must be first included in the animal protocol and approved by IACUC.
What are the risks of using contrast agents?
If the safety protocol and recommended guidelines are properly followed, there should be minimal risk to the operator associated with the use of MRI contrast agents.
When carrying animal studies consisting of contrast enhanced acquisitions, it is important to ensure that the dosage, administration route, and clearance considerations follow recommended guidelines and have received IACUC approval. Unlikely, but possible secondary effects after exposure to Gadolinium-based contrast agents include allergic reactions, headaches, nauseas, hives, vomiting… If the operator is exposed to MRI contrast agents, please contact the CILS Core Director ASAP.
How can the risks of MRI contrast agents be reduced?
The two key words are: research & communication! A mandatory safety training is required before using the MRI scanner. A detailed MRI Safety Protocol document can be downloaded BioRaft, and a summary can be found in the MRI room. There are additional resources available online including books, publications, videos…
If before, during, or after your experiments there are any questions or concerns that you would like to discuss, do not hesitate to reach out to the Core Facility staff. We are happy to help!
Other Questions
What if I still have questions or concerns?
If you have any additional questions or you would like further clarification, please contact CILS Core Director Guoxin Rong at g.rong@northeastern.edu.
MRI Applications
Other Instruments
Zeiss AxioZoom V16 (Upright)
NU user: free; Academia: $30/h; Industry: $50/h
Epifluorescence: DAPI, GFP, DsRed, Cy 5
Objective: 10×, 20×, 40×, 63×, 100×
Immunofluorescence labeled tissue or cell culture imaging
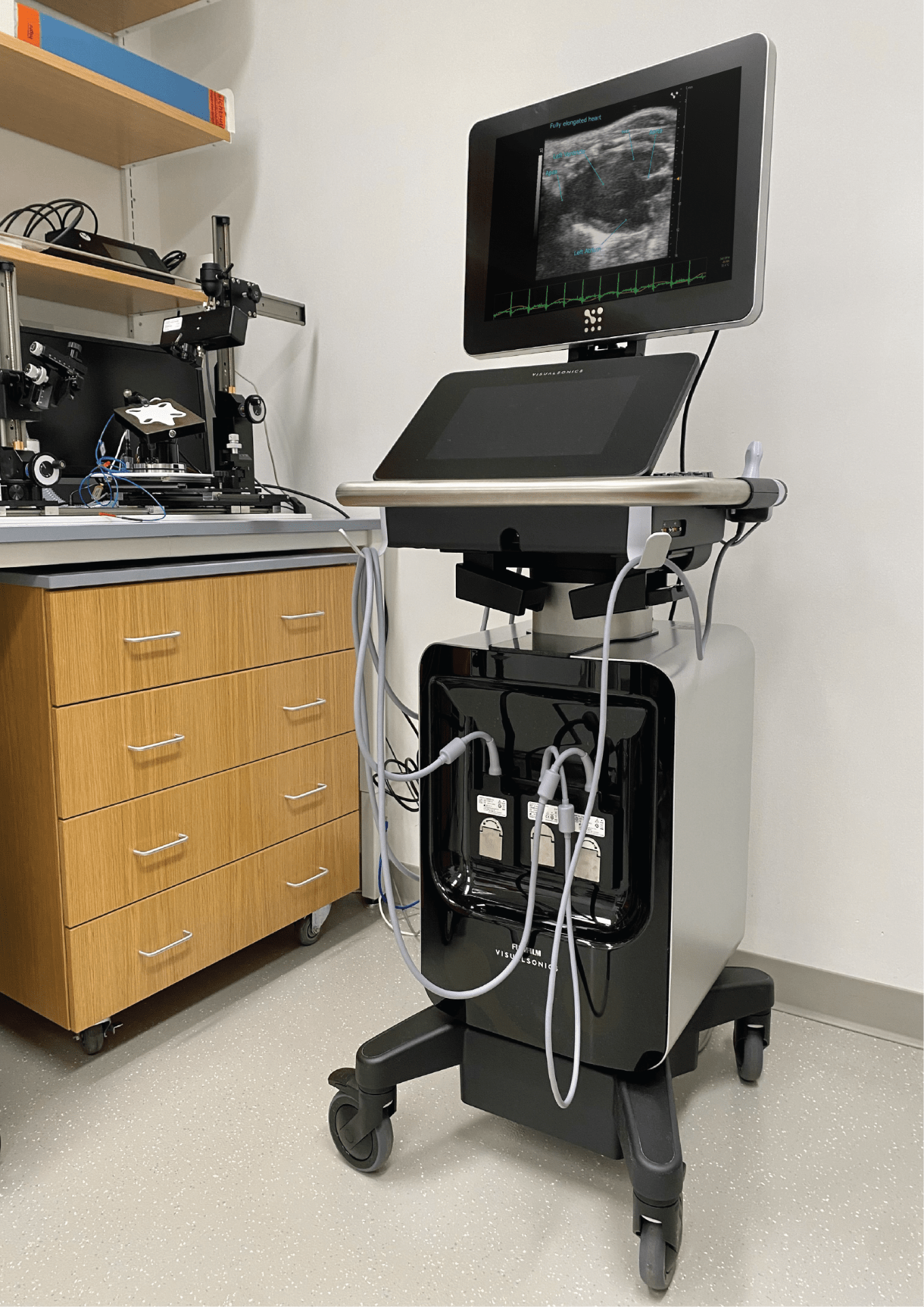
Visualsonics Vevo F2 Ultrasounds
NU user: $60/h; Academia: $110/h; Industry: $140/h
UHF57x, UHF29x, P10 transducer probes
Prerequisite: Bioraft Biosafety Training
Zeiss LSM 880 NLO (Upright)
NU user: $55/hr; Academia: $75/hr; Industry: $95/hr
Laser line: 405, 458, 488, 514, 561, 594, 640 nm
NIR Laser: tunable line (680-1300 nm); fixed line -1045 nm
Objective: 10×, 20×, 20×, 40×, 20×-CLARITY
Multi-photon imaging, Spectral Unmixing
Prerequisite: Bioraft Laser Safety Training Part I and II
Our facility
Interdisciplinary Science and Engineering Complex
ISEC 080, 805 Columbus Ave, Boston, MA, 02115